|
|
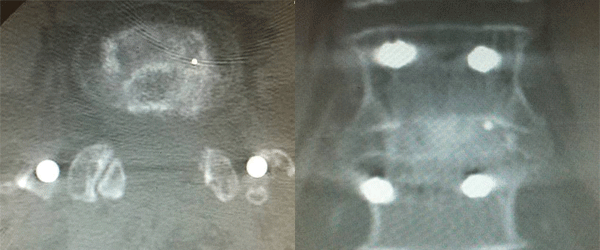
Kleiner Device Labs has announced full commercial availability of a new spinal bone graft delivery tool, the KG 1, featuring a patented design that facilitates less-invasive procedures and has been proven in clinical testing to reduce spinal fusion failure rates by 68%.
|
|
 |
|
Life Spine have announced that revenues for ProLift Expandable Spacer System grew by 493% for 2017 as compared to 2016. According to Life Spine, ProLift continues the company’s commitment of offering innovative Micro-Invasive procedural solutions to better improve patients’ lives.
|
|
 |
|
Spineology has announced the completion of the first post-market study cases using its recently FDA-cleared Duo Lumbar Interbody Fusion System. In October, the company announced the initiation of this prospective, post-market lateral interbody fusion study designed to evaluate patient outcomes.
|
|